Hey 2nd Yearites . !! As you are in 4th sem so first of all you might be thinking what chemical engineering is…. We are not definitely studying it the way we used to study chemistry in XI or XII standard,instead of that we are with subjects like Electrical Technology, TMMD, Solid mechanics. I’ll tell you why these subjects are important. As chemical engineer you will be able to come up with technical solutions for problems and issues in relation to process and product technology. In finding these solutions, chemical engineers work closely with experts from other specializations, taking into account the related economical, social, environmental and ethical aspects of the problem they’re dealing with. To understand it let us see one practical situation wherein you have task to build up a transportation pipeline from point A to point B and no qualified person is with you to help except one or two labourers. Your area of concern will be : 1) Angle of elevation of point A and point B from horizontal. 2) Diameter,length of pipe and which type of joints should be there? — So here comes Theory of machines and machine designing. 3) From what material pipe is to be made ? and what are the possibilities of failure of such a structure? If pipeline is to be made underground then the nature of soil needs to be understood to avoid corrosion problems— So here comes Chemical Engineering Materials. Finally let’s say you have carefully made all arrangements and the pipeline is ready to use. And as you switch on the pump, you may not see the fluid coming out from the pipe!!Then what will you do??? What is the problem??? Problem was that you may not used proper motor which will provide you exact power or you have purchased correct HP motor but it is consuming more power, then you will go and see whether my motor is delta or star connected because every connection has its advantages and disadvantages. So if you have studied electrical engineering basics then you would have easily identified the problem.
- Please don’t take any subject lightly or for the sake of getting marks. Strictly speaking what I think from my experience is that an engineer should have the know of basics of all disciplines of engineering. Now you may ask is coding and programming is going to help a chemie?? The answer is yes, because chemical process calculations are not as simple as solving an two variable equation and finding the answers. Practically any chemical process or even a small unit operation will have ‘n’ no. of variables so how you can solve them. So we need simulation software to handle these large no of variables. Now if you have basic “funda” clear in your mind that how it was designed and programmed.. You can handle the software much better than your colleague. But again I am not saying that you should be ‘phoodu’ in programming but basics are compulsory.
- So, Chemical or process engineering is an interdisciplinary science comprising elements of mechanical Engineering, chemistry and technical physics.
- The difference between chemical Engineering and process engineering lies in the emphasis of the degree course: while chemical engineers concentrate mainly on chemical processes, process Engineers deal primarily with the plants needed for this, their design and technical solution.
- For General Chemical Technology (GCT) please watch you tube animation videos, NPTEL lectures. Also you can distribute these topics among your friends and then have a discussion for e.g. one can prepare about paper and pulp industry , other one on sugar industry and discuss among themselves.
I hope you are clear what are the application and importance of these subjects in the life of chemical engineer.
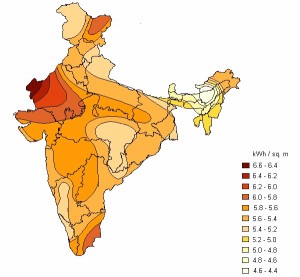
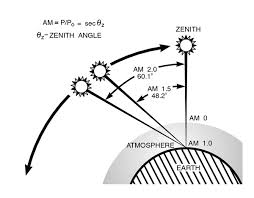
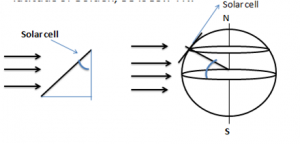
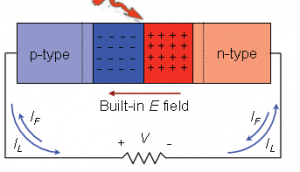
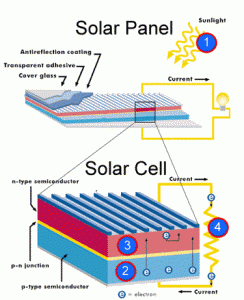
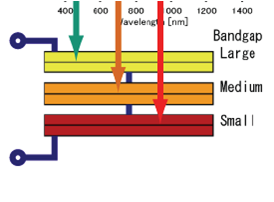
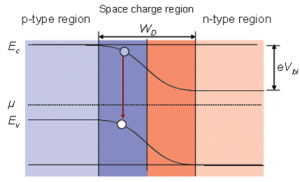
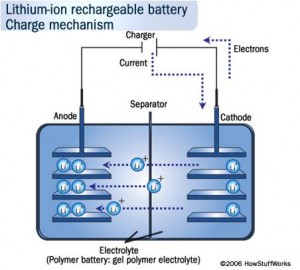
- For first yearites : Observe the chemical processes occurring in day to day life and analyze them. See corrosion problems , how thermodynamics is playing in your home kitchen, analyze how propulsion systems works , analyze how energy sources like batteries, fuel cells, solar cells works.
- Study alternate sources of energies such as bio diesel ( Jatropha seeds).Go through some basic outline of industries such as:Sugar industry, paper and pulp industry, soap and detergent industry….etc.
- If possible visit as many industries as possible.
- At the end of first year try to learn Microsoft Excel Software as much you can and C programming language in summer vacations.
- For second yearites.. We dont have core chemical engineering subjects in 2nd year… So in 2nd year have active participation in techfests of various colleges; this will give you experience, direction , boost up your confidence and will definately gives practical knowledge which helps when you will study core subjects such as (HTO, MTO, Thermo, CRE..etc)
- You can participate in events like chemical car competition and chemistry related quizzes in 2nd yearand then in 3rd year you can participate in heat exchanger event …(I am saying this because there is a separate topic on heat echanger in 5th semester,so it is better to participate in this event later on in 3rd year.) … Do as per your interest.
- Read magazines these are available in library, watch NPTEL lectures they are very good.
- There are some free online courses available online these are given by profs of MIT , Harvard, Oxford, Stanford.. So make use of them. Most recommended courses are EDX and Stanford university online course.
- My advice to you all is that don’t do industrial training in 2nd year.Instead go to your seniors, profs and work under them and learn as much you can from them. The best places for summer training in chemical engineering some are: IITs, IISC , ISER, NCL, ICT, CSMCRI etc and in Private colleges we have very good Nanotechnology Lab at DDIT college in Nadiad. and in our college itself we have summer training programme so apply for that .
Some preferred courses: EDX
- our energetic earth
- Introduction to water treatment
- Solar energy
- Introduction to solid state chemistry
- Thermodynamics
STANFORD ONLINE COURSE