“Fire breaks out in a building,” “Australia’s biggest forest fire ever rages.” We often hear about such devastative fire incidents in the media. We know that fire is dangerous and can cause severe damage and destruction and, at times, death. Since our earliest days, humans have sought to find out what fire is, how it starts, and what keeps it going.
Sometimes we might think that fire is a living thing! It moves, ‘eats’ things, and seems to breathe. The ancient Greeks thought it was one of four major elements, along with water, earth, and air. They could feel, see, and smell fire just like they could the earth, water, and air, but fire is something completely different.
Let us go on a journey to unveil the world of fire!
What is Fire? Which state is it? A solid or a liquid or a gas or plasma?
No, it is neither of them. Fire is just a perception of matter that is experienced by the eyes. Typically, fire results from a chemical reaction between oxygen in the atmosphere and a variety of fuels. When the volatile gases are hot enough, the compound molecules break apart, and the atoms recombine with the oxygen to form water, carbon dioxide, and other products. In other words, they burn, which results in a fire. The rising carbon atom is the reason for the production of light during the fire. Ignition temperature needs to be achieved for the combustion reaction to occur. During this reaction, the weak double bond of molecular oxygen gets converted into the stronger bonds of carbon dioxide and water, therefore, releasing energy, and this is the reason why fire is hot. The chemical reactions in a fire are self-perpetuating. The heat required by fuel is given by the heat of the flame itself, so as long as there is fuel and oxygen around it, the fire will continue.
FIRE TRIANGLE
The fire triangle is a triangle consisting of three components that help in the production of fire that are heat, oxygen, and fuel. Removal of any one of them will extinguish the fire. The alternative of the fire triangle is fire tetrahedron, which includes chemical reactions too, with all the other three components.
TYPES OF FIRE
- CLASS A- Fires involving ordinary combustibles such as wood, rubber, paper, cloth, and many plastics.
- CLASS B- Fires involving flammable gases such as gasoline, petroleum greases, tars, oils, oil-based paints, alcohols, solvents. It also includes combustible gases such as propane and butane.
- CLASS C- Fires involving energized electrical equipment such as computers, motors, transformers, and other appliances.
- CLASS D- Fires in combustible metals such as magnesium, titanium, zirconium, sodium, lithium, and potassium.
- CLASS K-Fires in cooking oils and greases such as animal and vegetable fats.
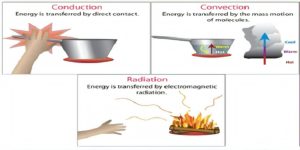
HOW FIRE SPREAD?
Once a fire has started, it grows through the transfer of heat energy from the flames. Heat energy transfers in three different ways-
- CONDUCTION- The heat from the fire spreads from molecule to molecule along the length of conducting materials. Materials that are good conductors absorb the heat from the fire and transfer it throughout the molecules of the substance.
- CONVECTION- It occurs in gases and liquids. It is the flow of fluid or gas from hot areas to colder areas. The heat of the fire raises the temperature of the air around it, which rises and spreads, which may burn the combustible materials.
- RADIATION- Heat of the Fire travels in the form of electromagnetic rays in air. Combustible materials can absorb the heat from the rays.
FIRE IN ZERO-GRAVITY
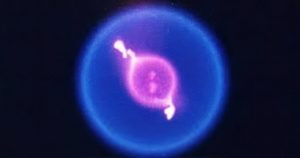
On earth, gravity determines how the flame burns. The product of combustion has more energy than the combustible substance and so moves around faster and takes up more space than the cooler air around them. Therefore, there is a buoyant force on them, which is higher than their weight. The hot gases in the flame are much warmer and less dense than the surrounding air, so they move upwards towards low pressure. This is why fire typically spreads upwards. However, in a zero-gravity region, there is no such thing as lighter or heavier air; thus, the fire heats the air, which just sits around the flame, causing it to burn slowly. This means the flame burns equally in all directions forming a globe instead of the flickering flame. Flames in the air can burn more slowly more coolly and with less oxygen because of which fire in space given the right conditions can expand in any direction as quickly as it can provide us to the nearby oxygen. The heat does not cause any rushing air or shockwaves. The cool thing found is that in space, combustion can happen with no visible flames. This phenomenon is demonstrated by the experiments conducted by NASA in the International Space Station, the Flame Extinguishment Experiment(FLEX). A more efficient combustion system that will not produce as much exhaust on earth can be made if these flames can be used as they burn cleaner.
FIRE SPREAD IN DIFFERENT SCENARIOS
- Fire in radioactive materials– Chernobyl incident was a nuclear accident in which radioactive material was present in the fire situation. Fire involving radioactive materials can result in widespread contamination. Radioactive particles can be carried easily by smoke plumes. Radiation includes alpha particles, which are extremely hazardous to people coming in contact with the fire because they can be inhaled and deposited in body tissues, where they can cause severe long term health effects.
- Fire in wood– Wood is a combustible material. Under the influence of heat, wood produces substances that react eagerly with oxygen, leading to the high propensity of timber to ignite and burn. Ignition and combustion of wood are mainly based on pyrolysis of cellulose and reactions of pyrolysis products with each other and with gases in the air, oxygen. When the temperature increases, cellulose starts to pyrolyze. The decomposition products either remain inside the material or are released as gases. Gaseous substances react with each other and oxygen, releasing a large amount of heat that further induces pyrolysis and combustion reactions.
Wood(C10H15O7)+heat —> Charred wood(C50H10O) + 10 CH2O(gas)
Forest fires include fire in a wood. Amazon forest fires and Bushfires in Australia are the major incidents, including the burning of wood. Fires in forests spread quickly due to the presence of combustible materials, which results in the realization of the fire triangle.
- Fire in oil- Oils are flammable materials which are less denser than water, so floats on it. Disaster due to fire in oils includes oil well fires. Oil well fires are oil or gas wells that have caught on fire and burn. Oil well fires can be a result of human actions, resulting in accidents, which can be a result of arson or due to natural events, such as lightning. These fires are more difficult to extinguish than regular fires due to enormous fuel supply to the fire. The significant incidents include Kuwait Oil Fires and Deepwater Horizon Explosion.
Conclusion
Fire is a perception of our eyes to the exothermic combustion reaction. This is part of the Mini Analysis Project “Study and analysis of the phenomenon of Fire and it’s practical Implications through Case Studies”
This was an introductory blog describing the true nature of fire!
So ending this with a sneak peek of case studies we are going to elucidate in further blogs:
- Chernobyl Nuclear Disaster
- Australian BushFires
- Amazon Rain Forests
Intriguing blogs about the same coming soon…
Stay tuned until then.
REFERENCES
- “Glossary of Wildland Fire Terminology” (PDF). National Wildfire Coordinating Group. November 2009. Retrieved 2008-12-18.
- ^ Schmidt-Rohr, K (2015). “Why Combustions Are Always Exothermic, Yielding About 418 kJ per Mole of O2“. Chem. Educ. 92
- “Iraq Fires erupt in large Iraqi oil field in south Compiled from Times wires © St. Petersburg Times published March 21, 2003”. Archived from the original on July 15, 2014.
- ^ “Hellfighters”. Archived from the original on 2014-07-14.
- https://science.howstuffworks.com/environmental/earth/geophysics/fire.htm
- https://science.howstuffworks.com/environmental/earth/geophysics/fire1.htm
- https://www.space.com/13766-international-space-station-flex-fire-research.html
Author: Hardik Khandelwal
TEAM CEV!!